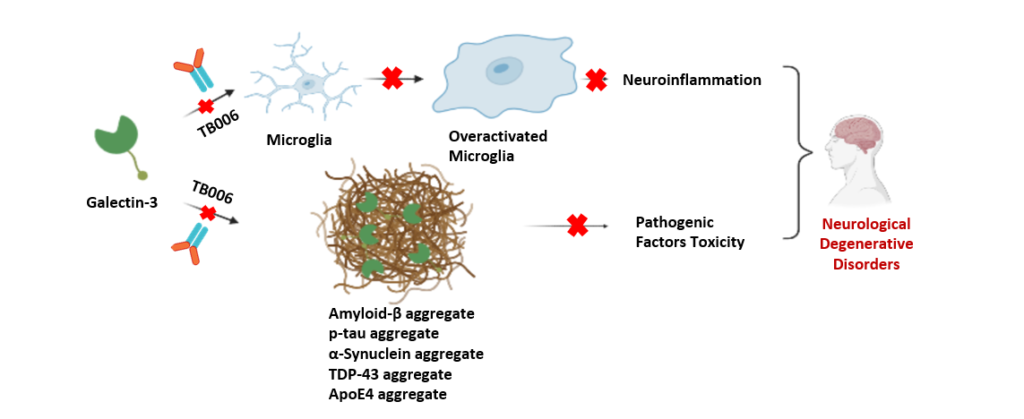
Low-complexity domains (LCDs) — flexible, disordered protein regions that drive neurodegeneration through pathological protein aggregation. A key focus is the LCD of Galectin-3, a protein increasingly implicated in diseases like Alzheimer’s and Parkinson’s.
Galectin-3’s N-terminal LCD acts as a molecular “sticky switch” that promotes aggregation in two main ways:
Self-association and scaffolding, forming oligomers that capture misfolded proteins like amyloid-beta or alpha-synuclein.
Liquid-liquid phase separation (LLPS), creating dense droplets that concentrate amyloidogenic proteins and accelerate toxic aggregate formation.
Beyond aggregation, Galectin-3’s LCD also fuels neuroinflammation by:
Activating microglia (the brain’s immune cells),
Creating a vicious feedback loop between aggregation and inflammation,
Directly amplifying inflammatory signaling via receptor clustering.
In essence, Galectin-3’s LCD is a critical driver of both protein aggregation and chronic inflammation—two central, self-reinforcing mechanisms in neurodegenerative disease progression.
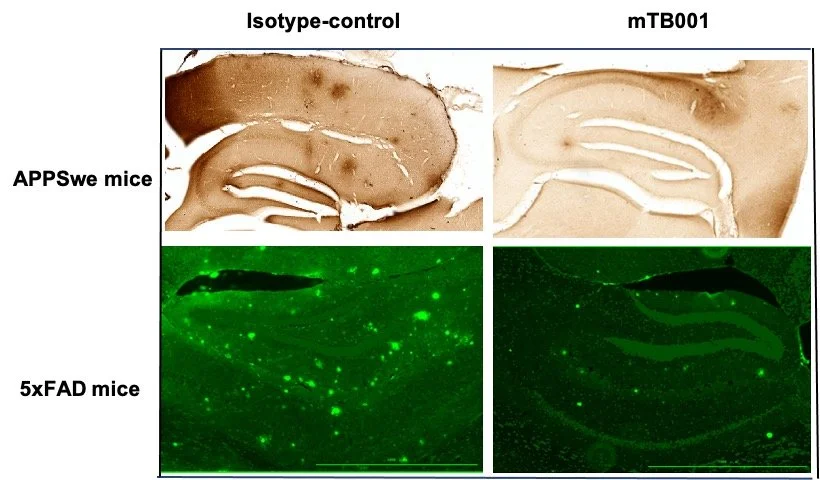
To learn more about TB006, you can also visit the site https://www.truebinding.com/ourscience